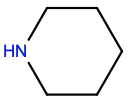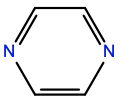MxMD Cosolvent Probes
The default cosolvent probes are shown in Table 1. These cosolvents were chosen because they are water-miscible and are known to mix evenly in MxMD simulations without phase separation [3]. Note that these probes are optimized for detecting protein hotspots. For DNA/RNA targets, you might want to use MxMD Custom Cosolvent Probes.
The MxMD workflow also supports immiscible probes. The allowed immiscible probes are listed in Table 2.
Each probe includes ten distinct pre-equilibrated solvent boxes that are used to assemble the initial cosolvent systems.
Table 1. Miscible cosolvent probes for MxMD. Default probes are acetonitrile, isopropanol, and pyrimidine (their names are highlighted in bold).
|
Name |
PDB Residue Type |
2D Structure |
Chemical Formula |
Mapping Role |
|
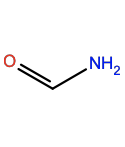
Acetaldehyde |
ACD |
|
C2H4O |
Polar; H-bond acceptor |
|
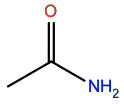
Acetamide |
ACM |
|
C2H5NO |
Polar; H-bond acceptor/donor |
|
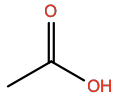
Acetic Acid |
ACY |
|
C2H4O2 |
Polar; H-bond acceptor/donor |
|
Acetone |
ACN |
|
C3H6O |
Polar; H-bond acceptor |
|
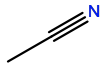
Acetonitrile |
CCN |
|
C2H3N |
Polar; amphipathic |
|
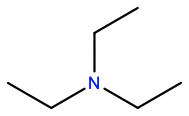
Diethylamine |
DME |
|
C4H11N |
H-bond donor; amphipathic |
|
Dimethylsulfoxide / DMSO |
DMS |
|
C2H6OS |
Polar; H-bond acceptor |
|
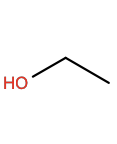
Ethanol |
EOH |
|
C2H6O |
Polar; H-bond donor |
|
Formamide |
ARF |
|
CH3NO |
Polar; H-bond acceptor/donor |
|
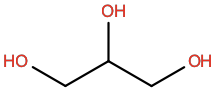
Glycerol |
GOL |
|
C3H8O3 |
Polar; H-bond donor |
|
Imidazole |
IMD |
|
C3H4N2 |
5-membered polar aromatic |
|
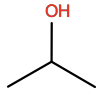
Isopropanol |
IPA |
|
C3H8O |
H-bond donor/acceptor; small hydrophobic |
|
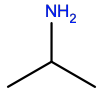
Isopropylamine |
ISP |
|
C3H9N |
H-bond donor; small hydrophobic |
|
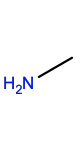
Methylamine |
NME |
|
C1H5N |
Small hydrophobic |
|
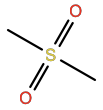
Methylsulfonylmethane/DMSO2 |
7SU |
|
C2H6O2S |
Polar; H-bond acceptor |
|
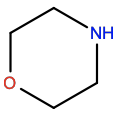
Morpholine |
6LR |
|
C4H9NO |
6-membrane; polar |
|
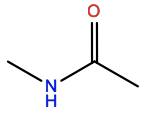
N-Methylacetamide |
NML |
|
C3H7ON |
Protein backbone-like |
|
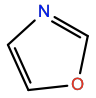
Oxazole |
OXZL |
|
C3H3NO |
5-membered aromatic; polar |
|
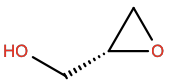
Oxiranemethanol |
OX9 |
|
C3H6O2 |
3-membered aliphatic; H-bond donor/acceptor |
|
Piperidine |
PIP |
|
C5H11N1 |
6-membered aliphatic; H-bond donor |
|
Pyrazine |
1P4 |
|
C4H4N2 |
6-membered aromatic |
|
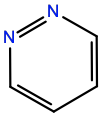
Pyridazine |
1P2 |
|
C4H4N2 |
6-membered aromatic |
|
Pyridine |
PYR |
|
C5H5N1 |
6-membered aromatic |
|
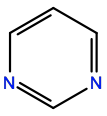
Pyrimidine |
P1R |
|
C4H4N2 |
6-membered aromatic |
|
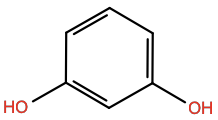
Resorcinol |
RCO |
|
C6H6O2 |
H-bond donor/acceptor; 6-membered aromatic |
|
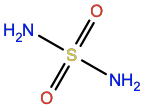
Sulfamide |
FUS |
|
H4N2O2S |
Polar; H-bond donor/acceptor |
|
Thiazole |
THZL |
|
C3H3NS |
5-membered aromatic |
|
Trifluoroethanol |
TFE |
|
C2H3F3O |
Polar; H-bond donor/acceptor |
|
Triethylamine |
TME |
|
C6H15N |
Small hydrophobic |
|
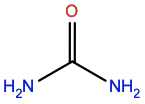
Urea |
URE |
|
CH4N2O |
Polar; H-bond donor/acceptor |
Table 2. Immiscible cosolvent probes for MxMD.
| Probe Name | PDB Residue Type |
|---|---|
| 1,3-Oxazinan-2-one | OX2 |
| 1-Methyl-1H-Indazole | MI1 |
| 1-Naphthol | 1NP |
| 1H-Indazole | LZ1 |
| 2-Cyanophenol | 0R0 |
| 2-Methyl-2H-Indazole | MI2 |
| 2-Naphthol | 2NP |
| 2H-Indazole | LZ2 |
| 3-Cyanophenol | 0R3 |
| 4-Cyanophenol | 0R4 |
| Androstenol | AND |
| Benzene | BNZ |
| Benzothiazole | BZL |
| Cyanobenzene | B1X |
| Furan | FUR |
| Isatin | ISN |
| Isobutane | ISB |
| Isonicotinonitrile | 4NN |
| Isoquinoline | ISQ |
| Isoquinolinone1 | IQ1 |
| Isoquinolinone2 | IQ2 |
| Nicotinonitrile | 3NN |
| Oxane | OXA |
| Oxirane | OXO |
| Phenol | IHP |
| Picolinonitrile | 2NN |
| Propyne | PYN |
| Quinoline | QNL |